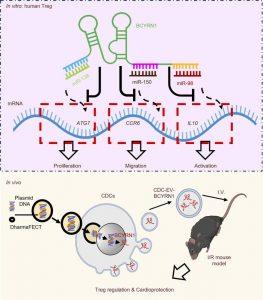
In a recent paper, researchers have developed a novel immunotherapy technique that dramatically improved healing after heart attacks in mice by accelerating the body’s production of anti-inflammatory immune cells (Figure 1). The findings hold promising implications not just for heart attack recovery, but potentially for treating a wide range of inflammatory diseases.
To combat this, the team zeroed in on regulatory T cells (Tregs)—specialized immune cells that suppress inflammation and keep immune responses in check. Previous approaches to boosting Tregs involved extracting them from patients, growing them in a lab, and reinfusing them—a process that takes weeks and is too slow to help during a heart attack.
The researchers discovered a new way to boost Tregs directly inside the body by using extracellular vesicles (EVs)—tiny sacs released by cells that can carry bioactive molecules through the bloodstream. They used a small RNA molecule called BCYRN1, found in high levels in a specific type of progenitor cell.
The EVs used in the study were derived from cardiosphere-derived cells (CDCs). These cells naturally produce BCYRN1, making them an ideal vehicle for this therapy.
Key Takeaways:
- Researchers have developed a fast-acting technique to increase anti-inflammatory regulatory T cells (Tregs) after a heart attack.
- The method uses extracellular vesicles carrying BCYRN1, a small RNA that boosts Treg numbers and suppresses damaging inflammation.
- Mice treated with this method had less heart damage, more immune protection, and better cardiac function post-heart attack.
- This technique could one day lead to new treatments for inflammatory conditions, including autoimmune diseases and transplant rejection.
With further development, this approach could mark a new era of rapid-response immunotherapy—where the body’s own cells are empowered to stop inflammation before it causes lasting harm.
Journal article: Liao, K., et al., 2025. Long non-coding RNA BCYRN1 promotes cardioprotection by enhancing human and murine regulatory T cell dynamics. Journal of Clinical Investigation.
Summary by Stefan Botha